Should red blood cell transfusion be immediately given to a cirrhotic patient with active upper gastrointestinal bleeding?
Introduction
Upper gastrointestinal bleeding (UGIB), mainly including variceal and non-variceal bleeding, can lead to a mortality of 2–15% (1). Transfusion of packed red blood cell (PRBC) is often required for the management of acute UGIB (2). Traditionally, if a patient presents with active UGIB, blood transfusion is immediately prescribed by the physicians. However, the appropriateness of this strategy remains uncertain. Herein, we attempted to discuss the optimal window of blood transfusion by showing a cirrhotic patient with acute UGIB who developed repeated hematemesis followed by blood transfusion.
Case presentation
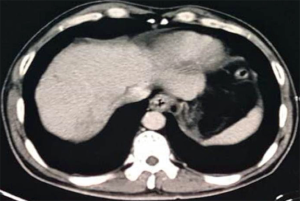
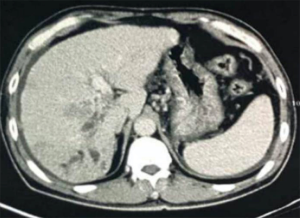
At 11:30 on February 27, 2018, a 57-year-old male was admitted to our department due to intermittent hematemesis and melena for about 12 days. He had a history of hepatitis B virus infection for 20 years and liver cirrhosis for 10 years. He underwent an endoscopic examination at his local hospital on February 11, 2018, which showed the evidence of esophageal varices with a maximum diameter of about 6 mm with a positive red-color sign and gastric varices, but did not undergo endoscopic therapy. He underwent contrast-enhanced computed tomography (CT) scans at his local hospital on February 22, 2018, which showed liver cirrhosis, ascites, and gastroesophageal varices (Figure 1), splenomegaly, a mass in the right hepatic lobe, portal vein tumor thrombosis (Figure 2), a fistula from hepatic artery to portal vein, and an ascending colonic wall swelling. Immediately after the present admission, he developed hematemesis with fresh blood of approximately 600 mL in volume. At that time, blood pressure was 110/87 mmHg, heart rate was 87 b.p.m., and oxyhemoglobin saturation was 100%. Laboratory tests demonstrated that red blood cell (RBC) was 3.27×1012/L (reference range: 3.8–5.1×1012/L), hemoglobin (Hb) was 128 g/L (reference range: 115–150 g/L), hematocrit (HCT) was 30.3% (reference range: 35–45%), prothrombin time (PT) was 14.4 s (reference range 11.5–14.5 s), and international standardization ratio (INR) was 1.14. PRBC 3 U and fresh frozen plasma 230 mL were urgently infused and terlipressin, somatostatin, esomeprazole, and ceftriaxone sodium were intravenously given. An urgent endoscopy was refused.
At 15:37 on February 27, 2018, he developed hematemesis again with fresh blood of approximately 600 mL in volume. He remained conscious, and blood pressure was 118/76 mmHg, heart rate was 86 b.p.m., and oxyhemoglobin saturation was 100%. Laboratory tests demonstrated that white blood cell (WBC) was 6.7×109/L (reference range: 3.5–9.5×1012/L), percentage of granulocyte (GR%) was 89.6%, RBC was 2.39×1012/L, Hb was 74 g/L, HCT was 22.1%, total bilirubin (TBIL) was 13.3 umol/L (reference range: 5.1–22.2 umol/L), direct bilirubin (DBIL) was 7.5 umol/L (reference range: 0–8.6 umol/L), alanine amino-transaminase (ALT) was 42.71 U/L (reference range: 9–50 U/L), aspartate amino-transaminase (AST) was 32.13 U/L (reference range: 15–40 U/L), alkaline phosphatase (AKP) was 115.83 U/L (reference range: 45–125 U/L), γ-glutamyl transpeptidase (GGT) was 209.10 U/L (reference range: 10–60 U/L), and albumin (ALB) was 30.5 g/L (reference range: 40–55 g/L). Child-Pugh score was 7 points. Except for terlipressin, somatostatin, esomeprazole, and ceftriaxone sodium, PRBC 3 U and fresh frozen plasma 270 mL were prescribed again.
At 19:10 on February 27, 2018, he developed hematemesis again with a volume of fresh blood vomited of approximately 200 mL. PRBC 3 U was prescribed again.
At 2:45 on February 28, 2018, he had hematemesis again with a volume of fresh blood vomited of approximately 100 mL. PRBC 2 U was prescribed again.
At 5:00 on February 28, 2018, he had hematemesis again with a volume of fresh blood vomited of approximately 200 mL. Laboratory tests demonstrated that WBC was 14.6×109/L, GR% was 88.6%, RBC was 2.47×1012/L, Hb was 77 g/L, HCT was 23.1%, and PLT was 97×109/L. PRBC 3.1 U was prescribed again.
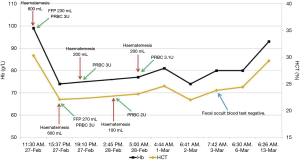
After that, the patient did not develop hematemesis again. On March 6, 2018, he underwent elective endoscopy and was treated with esophageal variceal band ligation. His condition was gradually improved (Figure 3).
iMDT discussion
Discussion among physicians from the General Hospital of Shenyang Military Area
Because acute UGIB will lead to a decrease of tissue perfusion after acute blood loss (3,4), the infusion of PRBC is often required. However, the timing of blood transfusion is still controversial. A UK study suggested that patients with acute UGIB who received blood transfusions had more frequent re-bleeding than matched patients who did not receive blood products and had a tendency towards increased mortality (5). A randomized controlled trial showed that “restrictive” transfusion (the hemoglobin threshold for transfusion was 7 g/L with a target range for the post-transfusion hemoglobin level of 7–9 g/L) significantly reduced the mortality compared with “liberal” transfusion (the hemoglobin threshold for transfusion was 9 g/L with a target range for the post-transfusion hemoglobin level of 9–11 g/L) (6). In 2013, a meta-analysis showed that restrictive transfusion could significantly decrease the incidence of death in patients with UGIB (7). Recently, a meta-analysis also suggested that a restrictive transfusion strategy should be associated with a reduction in mortality and re-bleeding in patients with acute UGIB (8). However, in geriatric patients, liberal transfusion strategies might produce better outcomes than restrictive transfusion strategies (9).
Currently, the American Association for the Study of Liver Diseases (AASLD) guideline recommends a restricted infusion of PRBC for patients with variceal bleeding (2). The Baveno VI consensus proposes that the target of Hb for infusion of PRBC should be 70–80 g/L (10). The 2015 UK guideline also suggest either excessive or insufficient blood transfusions leads to adverse events. As for patients with stable hemodynamics, the target of Hb should be adjusted to 70–80 g/L (11). The Chinese guideline also suggests that blood transfusion is required when Hb is less than 60–70 g/L (12).
Our patient had a Hb level of 99 g/L at the time of first hematemesis, which was higher than the threshold for a restrictive transfusion strategy (70 g/L). However, considering that UGIB was active, blood transfusion was prescribed. Unfortunately, he experienced repeated hematemesis followed by blood transfusion. Active UGIB did not appear until his Hb declined to a relatively low level. Thus, a liberal transfusion as a potential cause of repeated hematemesis should not be excluded.
Several issues regarding the management of this patient were further discussed as follows.
Should RBC transfusion be immediately given to a cirrhotic patient with active UGIB?
Expert opinion 1
In cirrhotic patients, a more liberal approach may be adopted especially in situations when there are derangement of clotting profile and thrombocytopenia.
Expert opinion 2
For most patients, I wait for the hemoglobin value, but it depends on the patient conditions, as well as the time span for receiving the blood test result. Some authors have suggested that massive bleeding could be treated by emergency transfusions (13). However, there is no clinical evidence to support this recommendation, probably because it would be difficult to include patients with severe bleeding in clinical trials. Another problem is the definition of massive bleeding, which is not clear. For practical purposes, I suggest to prepare PRBC when the patient has clinical signs of hemorrhagic shock, because in this situation it would be reasonable to begin the transfusion before the hemoglobin result. On the other hand, if the patient has no signs of hemorrhagic shock, I would wait for the lab test.
Expert opinion 3
Blood transfusion is not necessary immediately at the timing of acute variceal hemorrhage. The case had a blood pressure of 118/76 mmHg and heart rate of 86 b.p.m. It means that the patient is not under life-threatening hypotension. Emergent blood transfusion is not necessary. However, the vital signs should be closely monitored. Blood should be prepared in case of emergency.
Expert opinion 4
In my view, this is a case of massive bleeding, irrespective of its cause and, rather than PRBC, fresh whole blood would be indicated. Fresh whole blood is meant as whole blood less than 1-week storage but if possible less than 24 h kept at room temperature. This product is routinely used in West Africa where cirrhosis massive bleeding is frequent and related to the high frequency of chronic hepatitis B. This product has been promoted by the American surgeons for the treatment of battle field massive bleeding and is now recommended in civil massive bleeding in the USA. This product not only contains functional platelets but also plasma with all active clotting factors. Whole blood has been eliminated from the list of blood product available in many developed countries but this strategy needs to be revised for cases like the one presented. In such a case, the issue is not only O2 transport and availability but also containing the bleeding for which platelets and fresh plasma is necessary.
Is the Hb concentration obtained at the time of active gastrointestinal bleeding reliable and accurate?
Expert opinion 1
It is sometimes not reliable because of the haemo-concentration during the time of active bleeding. In addition, the genuine Hb level may be more reliable to be measured after the active bleeding has been stopped.
Expert opinion 2
Indeed, Hb concentration can vary at the first 24 hours after the bleeding, leading to some inaccuracy (14). Prior studies showed that Hb can vary after bleeding occurred during surgical procedures (15). However, it is the most used parameter to indicate PRBC transfusion for such patients.
Expert opinion 3
The initial Hb was 128 g/L. The normal Hb result is unreliable. Usually the Hb drops subsequently.
How many units of PRBC should be infused in this patient? (optimal dosage of PRBC)
Expert opinion 1
I would adopt 1–2 pints of packed cell every time.
Expert opinion 2
Actually, I would not prescribe a PRBC transfusion unless the patient developed another complication, such as hemorrhagic shock.
Expert opinion 3
The follow-up Hb was 74 g/L. The amount of blood transfusion would be 2–4 Taiwan blood units; it is about 1–2 American units to maintain a Hb level of about 80 g/L.
Conclusions
Massive hematemesis is not an indication for blood transfusion. A wait-and-see strategy of PRBC transfusion may be considered in a cirrhotic patient. Except for elder patients with cardiovascular or cerebrovascular abnormalities, blood transfusion may not be given until Hb concentration was less than 70 g/L. Instead, if he/she presented with active bleeding and had a Hb concentration of above 80 g/L, an emergency blood transfusion may elevate the portal pressure and induce the re-bleeding events.
Acknowledgements
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2018.08.02). Fernando Gomes Romeiro serves as an unpaid editorial board member of AME Medical Journal from Apr 2017 to Apr 2019. Xingshun Qi serves as an Editor-in-Chief of AME Medical Journal. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Antunes C, Copelin IE II. Gastrointestinal Bleeding, Upper. Treasure Island (FL): StatPearls Publishing; 2018.
- Garcia-Tsao G, Abraldes JG, Berzigotti A, et al. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology 2017;65:310-35. [Crossref] [PubMed]
- Gralnek IM, Barkun AN, Bardou M. Management of acute bleeding from a peptic ulcer. N Engl J Med 2008;359:928-37. [Crossref] [PubMed]
- Farrar FC. Management of Acute Gastrointestinal Bleed. Crit Care Nurs Clin North Am 2018;30:55-66. [Crossref] [PubMed]
- (UK) NCGC. Acute Upper Gastrointestinal Bleeding: Management. Acute Upper Gastrointestinal Bleeding: Management. National Institute for Health and Clinical Excellence: Guidance. London 2012.
- Villanueva C, Colomo A, Bosch A, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med 2013;368:11-21. [Crossref] [PubMed]
- Wang J, Bao YX, Bai M, et al. Restrictive vs liberal transfusion for upper gastrointestinal bleeding: a meta-analysis of randomized controlled trials. World J Gastroenterol 2013;19:6919-27. [Crossref] [PubMed]
- Odutayo A, Desborough MJ, Trivella M, et al. Restrictive versus liberal blood transfusion for gastrointestinal bleeding: a systematic review and meta-analysis of randomised controlled trials. Lancet Gastroenterol Hepatol 2017;2:354-60. [Crossref] [PubMed]
- Simon GI, Craswell A, Thom O, et al. Outcomes of restrictive versus liberal transfusion strategies in older adults from nine randomised controlled trials: a systematic review and meta-analysis. Lancet Haematol 2017;4:e465-e74. [Crossref] [PubMed]
- de Franchis R, Baveno VI. Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol 2015;63:743-52. [Crossref] [PubMed]
- Tripathi D, Stanley AJ, Hayes PC, et al. U.K. guidelines on the management of variceal haemorrhage in cirrhotic patients. Gut 2015;64:1680-704. [Crossref] [PubMed]
- Guidelines for the diagnosis and treatment of esophageal and gastric variceal bleeding in cirrhotic portal hypertension. Journal of Clinical Hepatology 2016;32:203-17.
- Hernandez-Gea V, Berbel C, Baiges A, et al. Acute variceal bleeding: risk stratification and management (including TIPS). Hepatol Int 2018;12:81-90. [Crossref] [PubMed]
- Meseeha M, Attia M. Esophageal Varices. Treasure Island (FL): StatPearls Publishing; 2018.
- Seaman BW, Ponder E. The Estimation And Control Of Post-Operative Dehydration, with the Aid Of Hemoglobin And Plasma Protein Determinations. J Clin Invest 1943;22:673-85. [Crossref] [PubMed]
Cite this article as: Bai Z, Guo X, Li H, Yu X, Deng J, Yuen MF, Romeiro FG, Yang SS, Allain JP, Qi X; Written on behalf of AME Liver Disease Collaborative Group. Should red blood cell transfusion be immediately given to a cirrhotic patient with active upper gastrointestinal bleeding? AME Med J 2018;3:83.