
Effect of en bloc esophagectomy and total meso-esophagectomy on esophageal cancer patients: a systematic review and meta-analysis
Introduction
Despite the progress in multiple treatment modalities, esophageal cancer is one of the most prevalent and deadly carcinomas worldwide (1,2). Surgical resection of the esophagus remains the mainstay of radical treatment (3). However, radical resection can be traumatic, and non-extended surgery might put the patient at a risk of non-radical resection. Thus, the optimal circumferential extent of surgical resection has not yet been determined.
Different modalities of esophagectomy have been developed in order to explore the optimal circumferential extent of surgery according to precision medicine (4,5). En bloc esophagectomy (EBE) was originally proposed by Logan as an extended esophageal surgery (6). For middle and lower esophageal cancer, besides the esophagus, EBE involves resection of the pericardium anteriorly, pleural surfaces laterally, the thoracic duct, and all other lymphoid tissues on the ventral side of the spine posteriorly (7). Notably, Cuesta et al. described an anatomic fascia between the descending aorta and carina called the meso-esophagus, similar to the mesorectum. Thus, total meso-esophagectomy (TME) has become a newly explored surgical method (8,9). During TME, vascular and lymphatic fatty tissue and nerves around the esophagus are removed as a single anatomical unit ending at the level of the azygos vein superiorly. The meso-esophagus with the tracheoesophageal sulcus and recurrent laryngeal nerve (RLN) lymph nodes (LNs) are dissected along the spine to the thoracic outlet. A previous study described that extensive resections could be accompanied by high postoperative morbidity as well as superior long-term oncological outcomes (10). As reported by several studies, minimally invasive esophagectomy (MIE) can be a safe and feasible treatment modality for esophageal cancer, and the integration of EBE and TME with this advanced technique may shed a light on the surgical treatment of esophageal cancer (11).
Therefore, based on the results of randomized controlled trials (RCTs), we conducted the present systematic review and meta-analysis to evaluate the outcomes of EBE and TME, as well as the long-term prognosis of the 2 surgeries. We present the following article in accordance with the PRISMA reporting checklist (available at https://amj.amegroups.com/article/view/10.21037/amj-22-38/rc).
Method
Search strategy
Several electronic databases were used to conduct the search strategy, including PubMed, Cochrane Library, Web of Science, and Embase, and the index date was up to March 1st, 2022. The search keywords included “esophageal cancer”, “en bloc esophagectomy”, and “meso-esophagectomy”. The references of the screened and selected studies were also manually searched in the databases and assessed. The details of the search strategy and results are listed in Tables S1,S2. Citation searches of the eligible studies and reviews were also manually performed for potential related articles.
Study selection
Articles were identified by 2 reviewers (FS, HJ) independently according to the inclusion and exclusion criteria. The inclusion criteria were as follows: (I) clinical studies that enrolled patients with histologically or cytologically confirmed esophageal carcinoma; (II) studies that used EBE or TME as the mainstay of radical curative therapy; (III) studies comparing EBE or TME with other surgeries for patients with esophageal carcinoma; (IV) studies reporting at least one of the following outcomes: operation time, intraoperative blood loss, LN dissection, R0 resection rate, postoperative morbidity and mortality, overall survival (OS), and disease-free survival (DFS). The exclusion criteria were as follows: (I) single-arm studies; (II) non-clinical studies.
Data extraction
Two authors (FS, HJ) reviewed the studies independently and extracted the continuous and discontinuous data from the studies. Characteristics of the studies including first author, publication year, research centers, methodology (study design, randomization, and allocation), sample size, demographic characteristics, treatment modality, and short-term and long-term outcomes were collected. The sample size, mean, and standard deviation (SD) were collected for continuous variables such as age, operation time, blood loss, and the number of LNs acquired. If the SD was not available in the literature, then the SDs would be calculated according to the guideline of the Cochrane handbook, while sample size and number of events were collected for discontinuous data such as sex distribution and R0 resection (12). As for OS and DFS, the rate at every time point was estimated from Kaplan-Meier curves with Engauge Digitizer (version 11.1). Sample size and cases at risk at several time points were also documented.
Evaluation of quality
The quality of the selected studies was evaluated by 2 reviewers (FS, HJ) with the Cochrane risk of bias tool for RCTs. The tool contains the following 7 key items: (I) randomization; (II) allocation concealment; (III) participant blinding; (IV) outcome assessment blinding; (V) incomplete outcome data; (VI) bias in reporting; and (VII) other bias. Each study was graded into 3 levels for each item as “low bias risk”, “unclear bias risk”, and “high bias risk” according to the reviewers.
Statistical analysis
Before comparison, the units of the outcomes were converted into standard international units. The software Review Manager 5.4 was used to calculate every outcome for the meta-analysis. For short-term outcomes, the risk ratio (RR) was used for the assessment of discontinuous data, and the mean difference (MD) was used for continuous data. The log-rank observed minus expected events (O-E) and the log-rank variance (V) were derived from Kaplan-Meier curve data and follow-up data (13). Survival benefits were recorded as a hazard ratio (HR) calculated from O-E and V. The 95% confidence interval (CI) was used to estimate the population parameter. A value of P<0.05 was considered statistically significant. The short-term and long-term results were shown in forest plots and funnel plots containing no less than 10 studies to avoid potential publication bias, which was assessed using Begg’s rank correlation test and Egger’s test in STATA (version 16.0).
I2 and Chi squared were derived from the results to evaluate heterogeneity between the studies. The P value was interpreted as follows: in the analysis of short-term outcomes, if I2≤50% or could be adjusted by excluding several heterogeneous studies, then we considered the heterogeneity between studies as not important and the fixed effects model with the Mantel-Haenszel method was used in the secondary analysis. If I2>50%, there was moderate or greater heterogeneity, and the random effects model with the Der-Simonian and Laird method was used for further analysis. The degree of heterogeneity was judged in terms of P value: 0.01<P≤0.05 indicated moderate heterogeneity and P≤0.01 indicated substantial heterogeneity. High heterogeneity may greatly harm the level of evidence grade, except that all of the studies showed a consistent direction in the forest plot.
Results
Search results
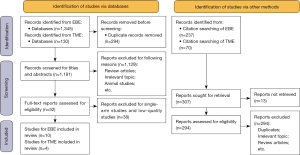
The search strategy yielded 1,475 and 307 articles from databases and citation searching respectively, and 294 duplicate records were removed with Endnote X9. Of the remaining 1,181 articles, 1,129 were excluded for reasons including irrelevant topic, review articles, and commentary articles. After evaluating the full text, 38 further exclusions were made according to the inclusion and exclusion criteria. Finally, fourteen studies met the inclusion criteria and were included in the meta-analysis (flow diagram shown in Figure 1).
Study characteristics
The publication centers of the included studies, which were published between 1993 and 2021, were in Europe (n=5) (14-18), North America (n=4) (19-22), and Asia (n=5) (23-27). Five of the included studies were published in the recent 5 years, while 2 were published before 2000. In 10 studies of EBE, patients in the control groups received transhiatal esophagectomy (THE) in eight studies and standard resection in 2 groups, while non-TME was performed in the control groups of four TME studies. In ten studies (14-22,25), there were 892 patients in the control groups and 1,105 patients received EBE in the study groups. In the remaining four studies (23,24,26,27), 566 cases of TME were performed compared with 553 cases of standard esophagectomy. A brief summary of the included studies is shown in Table 1.
Table 1
| Study | Country | Histology (SCC/AC/others) | Treatment strategy | Participants | Study design | Outcomes |
|---|---|---|---|---|---|---|
| Grotenhuis et al. 2010 (14) | Netherlands | 46/86/– | Transthoracic EBE with extended lymphadenectomy | 132 | RCT | Gender, age, operation time, LN acquired, R0 resection, postoperative complication, OS and DFS |
| 12/78/– | THE surgery | 90 | ||||
| Rizzetto et al. 2008 (19) | USA | –/40/– | Neoadjuvant therapy followed by EBE | 40 | RCT | Gender, age, LN acquired and OS |
| –/18/– | Neoadjuvant therapy followed by THE | 18 | ||||
| Donohoe et al. 2012 (15) | Ireland | Unknown | Transthoracic EBE | 438 | RCT | LN acquired, R0 resection, postoperative complication and OS |
| 8/24/– | THE surgery | 32 | ||||
| Yekebas et al. 2006 (16) | Germany | 79/– /– | Transthoracic EBE with two-field lymphadenectomy | 79 | RCT | Gender, LN acquired and DFS |
| 41/–/– | THE with en bloc lymphadenectomy | 41 | ||||
| Corsini et al. 2021 (20) | USA | –/133/– | En bloc transthoracic Ivor Lewis esophagectomy | 133 | RCT | Gender, age, operation time, blood loss, LN acquired, R0 resection, postoperative complication and DFS |
| –/471/– | Modified transthoracic Ivor Lewis esophagectomy | 471 | ||||
| Fu et al. 2012 (23) | China | Unreported | Thoracoscopic TME | 45 | RCT | Gender, age, operation time, blood loss, LN acquired and postoperative complications |
| Thoracoscopic esophagectomy | 61 | |||||
| Junhui et al. 2017 (24) | China | 122/10/– | Thoracoscopic TME | 132 | RCT | Gender, age, operation time, blood loss, LN acquired, postoperative complication and OS |
| 121/9/– | Mckeown open esophagectomy | 130 | ||||
| Fujiwara et al. 2016 (25) | Japan | 31/1/2 | THE plus transthoracic EBE | 34 | RCT | Gender, age, operation time, blood loss, LN acquired, R0 resection and postoperative complication |
| 21/2/– | Transhiatal subtotal esophagectomy | 23 | ||||
| Hagen et al. 1993 (21) | USA | 4/10/16 | EBE with gastrectomy | 30 | RCT | Gender, age and OS |
| 13/10/16 | Transhiatal esophago-gastrectomy | 39 | ||||
| Hulscher et al. 2002 (17) | Netherlands | –/114/– | Transthoracic EBE with extended lymphadenectomy | 114 | RCT | Gender, age, LN acquired, R0 resection, postoperative complication, OS and DFS |
| –/106/– | THE with en bloc lymphadenectomy | 106 | ||||
| Johansson et al. 2004 (18) | Sweden | –/27/– | Transthoracic EBE | 27 | RCT | Gender, age, LN acquired, R0 resection and OS |
| –/22/– | THE | 22 | ||||
| Lin et al. 2021 (26) | China | 249/– /– | TME | 249 | RCT | Gender, operation time, blood loss, postoperative complication, OS and DFS |
| 249/–/– | Non-total meso esophagectomy | 249 | ||||
| Altorki et al. 1997 (22) | USA | 38/90/– | EBE | 78 | RCT | R0 resection, postoperative complication and DFS |
| Standard esophageal resection | 50 | |||||
| Akiyama et al. 2018 (27) | Japan | 84/2/1 | TME | 87 | RCT | Gender, age, operation time, blood loss, LN acquired and postoperative complication |
| 51/–/2 | Non-meso esophagectomy | 53 |
EBE, eb bloc esophagectomy; TME, total meso-esophagectomy; THE, transhiatal esophagectomy; RCT, randomized controlled trial; LN, lymph nodes; OS, overall survival; DFS, disease-free survival.
Bias assessment
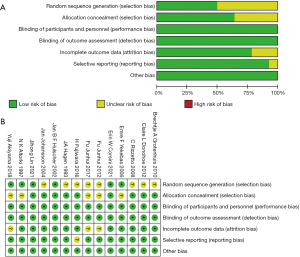
According to the Cochrane Library Handbook, performance biases, detection biases, or other biases were not observed in the studies. The main possible biases focused on selection bias and attrition bias. More than half of the studies did not describe the precise method for random sequence generation, and the allocation method was unable to be evaluated in five studies. Attrition bias in three studies and reporting bias in one study showed an unclear risk. The detailed biases of the included studies are shown in Figure 2, and the comprehensive risk of bias that affected the results was fairly low.
Demographic characteristics
The demographic characteristics of gender and age were collected. Eight EBE studies and four TME studies included statistics on gender distribution (14,16-21,23-27), and the gender distribution did not differ in the experimental group or control group [OR =0.90 (0.73, 1.12), P=0.34] but showed heterogeneity from subgroup differences and within-group differences. No significant difference was found in the subgroup analysis (Figure 3A). The age of participants in seven EBE studies and three TME studies were concluded (14,17-21,23-25,27). Although substantial heterogeneity existed, we observed that there were more younger participants in the experimental group [MD =−4.27 (−4.92, −3.63), P<0.001] (Figure 3B). This difference mainly came from the EBE subgroup [MD =−5.21 (−5.90, −4.52), P<0.001], but the TME subgroup showed an opposite result [MD =1.93 (0.15, 3.70), P=0.03].
Operation outcomes
The operation outcomes mainly included operation time, blood loss during surgery, the number of LNs dissected, and the R0 resection rate.
Operation time
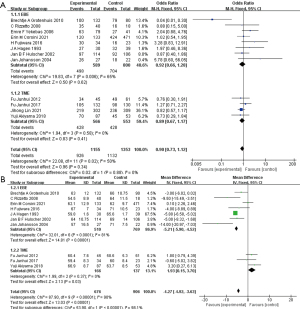
Operation time was included in seven studies and involved 2027 patients (Figure 4A) (14,20,23-27). As heterogeneity was obvious between the studies (I2=99%, P<0.001), we used the random effects model for the comparison. A different effect on the operation time was observed in the 2 subgroups. EBE had a borderline longer operation time [MD =101.44 (−1.66, 204.55), P=0.05], while TME had a shorter operation time [MD =−32.37 (−65.12, 0.37), P=0.05]. Although heterogeneity was substantial in both subgroups (I2=98%, P<0.001 for EBE; I2=99%, P<0.001 for TME), studies in the EBE subgroup unanimously indicated a longer operation time for EBE.

Intraoperative blood loss
The I2 was higher than 50% between the six included studies (I2=94%, P<0.001), and as a result, we used the random effects model in the analysis of blood loss during surgery (Figure 4B) (20,23-27). A diametrically opposite effect of EBE and TME on blood loss was found, so the combination of the 2 surgeries was not considered. There was significantly greater blood loss in EBE compared with the control group [MD =56.29 (14.80, 97.77), P=0.008; I2=0%, P=0.44]. The same trend of less blood loss was found in the four studies on TME [MD =−74.03 (−96.69, −51.38), P<0.001], regardless of the heterogeneity (I2=94%, P<0.001).
LNs dissected
The comparison of dissected LNs included twelve studies involving 2,765 patients. Eight studies involving 1,759 patients were on EBE, while four studies involving 1,006 patients were on TME (Figure 4C) (14-20,23-27). The experimental groups had more dissected LNs than the control groups [MD =11.63 (6.11, 17.15), P<0.001], and the random effects model was used due to substantial heterogeneity (I2=97%, P<0.001). All the studies showed a unanimous effect in the experimental group. Although the difference in the TME subgroup was not significant, both subgroups have substantial within-group heterogeneity (I2=88%, P<0.001 for EBE; I2=99%, P<0.001 for TME) and had a greater number of LNs dissected than the standard resection [MD =14.39 (9.79, 19.00), P<0.001 for the EBE group; MD =6.80 (−2.76, 16.35), P=0.16 for the TME group].
R0 resection rate
In the TME subgroup, only Lin et al. described that non-R0 resection was one of the exclusion criteria (26). As other studies did not mention the R0 resection rate, we only compared the R0 rate of EBE with the control group. The heterogeneity was significant between the six included studies with 1,701 patients (I2=61%, P=0.02), so the fixed effects model was used for analysis (Figure 4D) (14,15,17,20,22,25). EBE had a higher R0 resection rate [RR =1.06 (1.02, 1.11), P=0.002].
Postoperative outcomes
The evaluated postoperative outcomes included pulmonary complications, RLN injury, anastomotic complications, and postoperative mortality.
Pulmonary complications
Pulmonary complications mainly refer to postoperative pneumonia, respiratory failure, pulmonary embolism, and atelectasis pleural effusion requiring drainage. Six studies on EBE and four studies on TME were reviewed in this analysis (Figure 5A) (14,15,17,20,22-27). Due to high heterogeneity (I2=60%, P=0.008), the random effects model was used, and there was no significant difference in pulmonary complications postoperatively [RR =1.13 (0.89, 1.43), P=0.32]. Additionally, no significant difference in the incidence of pulmonary complications was found in the subgroup analysis [RR =1.22, (0.85, 1.75), P=0.27 and I2=71%, P=0.004 for the EBE subgroup; RR =0.97 (0.79, 1.18), P=0.75 and I2=0%, P=0.98 for the TME subgroup].

RLN injury
Symptoms described as vocal cord paralysis, RLN palsy, and hoarse voice were involved in the analysis. The included studies were the same as those on pulmonary complications and involved 2,707 patients (Figure 5B) (14,15,17,20,22-27). We observed a heterogeneous result with no difference in the incidence of RLN injury as determined by the random effects model [RR =1.08 (0.52, 2.22), P=0.84; I2=67%, P=0.001). Both EBE (RR =0.76 (0.27, 2.13), P=0.60) and TME [RR =1.77 (0.56, 5.60), P=0.33] were not associated with a higher risk of postoperative RLN injury.
Anastomotic complications
Postoperative anastomotic complications mainly included anastomotic leakage. Six RCTs on EBE and four RCTs on TME were included (Figure 5C) (14,15,17,20,22-27). Considering that heterogeneity between and within groups was minor (I2=0%, P=0.87 for total; I2=0%, P=0.85 for the EBE subgroup; I2=0%, P=0.46 for the TME subgroup), the fixed effects model was used. No significant difference was observed in the incidence anastomotic complications [RR =0.79 (0.60, 1.05), P=0.10], and a similar result was obtained in the subgroup analysis [RR =0.86 (0.58, 1.27), P=0.45 for the EBE subgroup; RR =0.73 (0.50, 1.08), P=0.12 for the TME subgroup].
Postoperative mortality
Eight studies and 2,339 participants were included in the comparison of postoperative mortality (Figure 5D) (14,15,17,20,22,25-27). As no significant heterogeneity was observed in the comparison or subgroup analysis (I2=0%, P=0.96 for the global analysis; I2=0%, P=0.93 for EBE; I2=0%, P=0.46 for TME), the fixed effects model was used. Postoperative mortality was not significantly different between the experimental group and control group in the global analysis or subgroup analysis [RR =1.15 (0.71, 1.84), P=0.57 for the global analysis; RR =1.17 (0.72, 1.91), P=0.52 for the EBE subgroup; RR =0.77 (0.10, 6.10), P=0.81 for the TME subgroup].
Long-term outcomes
The main indicators for long-term outcomes were OS and DFS. Patients in EBE studies were further grouped into early stage and advanced stage by TNM stage and LN status according to postoperative pathology. The pathological stage in the study by Hagen et al. was different from others, and it was considered as 2 studies in the advanced subgroup analysis, with Hagen-1 for intermediate stage and Hagen-2 for late stage (21).
OS
Seven studies provided the OS information of 1,364 patients (Figure 6A) (14,17-19,21,24,26). Heterogeneity was significant between studies (I2=58%, P=0.03), and OS was better in the extended surgical group than in the standard group [HR =0.79 (0.66, 0.94), P=0.008]. In the subgroup analysis, the EBE group showed moderate heterogeneity (I2=62%, P=0.03) and without significant superiority in OS [HR =0.82 (0.65, 1.03), P=0.08]. The TME group showed similar heterogeneity (I2=69%, P=0.07) and significantly better OS [HR =0.74 (0.55, 0.98), P=0.04].
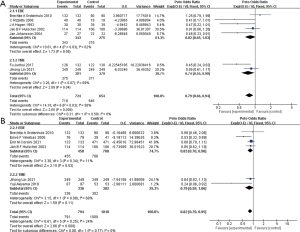
DFS
Six studies included the DFS data of 1,799 patients (Figure 6B) (14,16,17,20,26,27). The results showed a better DFS in the experimental group [HR =0.82 (0.70, 0.95), P=0.008], with subtle heterogeneity (I2=24%, P=0.25). In the EBE subgroup, the DFS was better than that in the control group [HR =0.83 (0.70, 0.98), P=0.03], with little heterogeneity (I2=11%, P=0.34). On the other hand, there was no strong positive result in the TME subgroup [HR =0.79 (0.59, 1.06), P=0.11; I2=68%, P=0.08].
Long-term outcomes for early-stage patients
The long-term outcomes of EBE were further analyzed. The analysis of OS included 2 studies and 36 subjects (Figure 7A) (19,21). Survival after EBE was longer than after regular surgery [HR =0.31 (0.10, 0.96), P=0.04; I2=0%, P=0.36]. As for DFS, the data of 351 patients from 3 RCTs were extracted (Figure 7B) (14,16,20), and no DFS benefit was observed in the EBE group or the control group [HR =0.71 (0.41, 1.21), P=0.21; I2=0%, P=0.99].
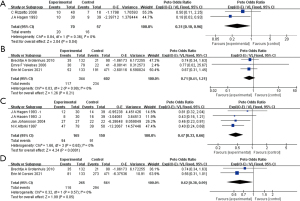
Long-term outcomes for advanced-stage patients
Similarly, the long-term outcome data of late-stage patients was collected. Three studies with 185 patients were analyzed (Figure 7C), and we found that EBE surgery resulted in significantly better survival than non-EBE surgery [HR =0.47 (0.33, 0.66), P<0.001; I2=0%, P=0.65]. DFS of 410 subjects with advanced disease from 2 studies was compared (Figure 7D) (14,20), and a positive significant result was derived from the data [HR =0.62 (0.38, 0.99), P=0.05; I2=0%, P=0.57].
Evaluation of publication bias
The evaluation of gender distribution and LN dissection included most studies, so funnel plots based on the meta-analysis of these 2 indicators were generated (Figure 8). The figures showed that there may be substantial publication bias resulting from a delay or lack of publication of non-statistically significant small RCTs. In addition, to determine possible bias in detail, Begg’s test and Egger’s test were performed for the other outcomes where no more than 10 articles were included (Table S3). There was no evidence to suggest the presence of publication bias in most outcomes except for age, operation time, and LN dissection.

Discussion
Based on the results of 14 RCTs, we performed this systematic review to investigate the clinical outcomes of EBE and TME surgery. To our knowledge, this is the first meta-analysis evaluating the efficacy of EBE as well as the safety and feasibility of TME. In the study, we found that EBE was accompanied by more intraoperative blood loss, more LNs removed, a higher R0 resection rate, as well as better long-term OS and DFS in both early- and advanced-stage patients, but there was no significant difference in postoperative complications or the incidence of mortality. As for TME, less intraoperative blood loss, shorter operation time, more LNs acquired, and lower HR for OS and DFS were observed. The studies did not compare the efficacy of EBE and TME directly. Considering the control group as a reference, the 2 surgeries were further compared. We concluded that TME has a shorter operation time, less intraoperative bleeding, and less dissected LNs compared with EBE, while the postoperative outcomes of the 2 surgeries are comparable. However, the results of long-term prognosis are hard to compare through the articles included.
In this review, no survival benefits were observed. This may be partially because the EBE group had a greater proportion of subjects at the advanced stage (16,17,19,25), and this imbalance of disease staging between groups could harm long-term efficacy in terms of survival benefits. After dividing into subgroups to perform comparisons, a lower HR in both early- and advanced-stage patients was observed. We also observed that not all patients in the EBE group achieved R0 resection, which is different from what is assumed, and may partially result from the inclusion of patients at stage III and IV. The R0 resection rate indicated the need for comprehensive treatment. Besides, no significant difference in postoperative complications was observed, and whether this resulted from the feasibility of EBE or the unbalanced distribution of demographic characteristics between groups needs further investigation.
The lower blood loss and shorter operation time found for TME may be due to the removal of the esophagus and its mesangial structure as a single unit according the anatomical gap, which may reduce vessel injury, ameliorate surgical field, improve LN dissection, and optimize the learning curve. This entire unit excision is not associated with a higher incidence of postoperative pulmonary complications, anastomotic complications, RLN injury, or mortality (26,27). Notably, as a newly developed surgical technique, the practice of TME is not widespread, and as a result, the number of studies investigating TME is limited, which is possibly responsible for some of the negative outcomes such as for DFS.
Interestingly, in several studies included in this meta-analysis, a thoracoscopic or laparoscopic surgical approach was used for part of the EBE surgery and all of the TME surgery (23-27). It is known that MIE is a surgical approach resulting in less trauma, enhanced recovery, and improved quality of life, with comparable oncological results (28-30). Besides, MIE is an option even for advanced esophageal cancer patients (31,32). However, considering the blood loss and operation time of EBE, combination with EBE could be highly technically demanding, which may limit its widespread application. On the other hand, several studies have proven the feasibility of MIE combined with TME (23,24,26,33), considering the effect of its learning curve. Such combination has the potential to become a mainstream procedure for esophagectomy in the future.
There are several limitations in our meta-analysis. Firstly, there are some potential biases in the study. The included studies were mostly retrospective, and the assessment found that publication bias could not be completely avoided. Besides, the risks of some studies, especially in selection bias and attrition bias, were unclear. Four out of 14 studies included in the meta-analysis were small-sized studies, which could limit the extrapolation of their conclusions. Another limitation is that several outcomes were fairly heterogeneous, probably resulting from the wide spread of research units around the world, which likely reduces the reproducibility of conclusions in particular populations. Notably, we included 2 studies in the meta-analysis that were published in Chinese, found in the Web of Science database. Considering that China is an area with a large burden of esophageal cancer (2), articles in languages other than English, especially in Chinese, were not rejected for further evaluation.
Conclusions
In this systematic review and meta-analysis, we found that EBE results in higher blood loss, longer operation time, and better long-term outcomes without increasing the incidence of postoperative complications. The operation time was shorter and bleeding was less than TME surgery, which could also improve the surgical and oncological efficacy. In conclusion, EBE is a safe and feasible extended esophagectomy method and TME is a promising innovative esophageal surgery, which is associated with better surgical outcomes in clinical practice. Further well-designed prospective RCTs are required to verify the results of this meta-analysis.
Acknowledgments
Funding: This work was supported by the National Natural Science Foundation of China (No. 81400681) and China Postdoctoral Science Foundation Grant (No. 2018M631394). The funding agencies had no role in study design, collection and analyses of data, decision to publish, or manuscript preparation.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://amj.amegroups.com/article/view/10.21037/amj-22-38/rc
Data Sharing Statement: Available at https://amj.amegroups.com/article/view/10.21037/amj-22-38/dss
Peer Review File: Available at https://amj.amegroups.com/article/view/10.21037/amj-22-38/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://amj.amegroups.com/article/view/10.21037/amj-22-38/coif). YXS serves as the Editor-in-Chief of AME Medical Journal. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Napier KJ, Scheerer M, Misra S. Esophageal cancer: A Review of epidemiology, pathogenesis, staging workup and treatment modalities. World J Gastrointest Oncol 2014;6:112-20. [Crossref] [PubMed]
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021;71:209-49. [Crossref] [PubMed]
- Short MW, Burgers KG, Fry VT. Esophageal Cancer. Am Fam Physician 2017;95:22-8. [PubMed]
- Jung MK, Schmidt T, Chon SH, et al. Current surgical treatment standards for esophageal and esophagogastric junction cancer. Ann N Y Acad Sci 2020;1482:77-84. [Crossref] [PubMed]
- van Rijswijk AS, Hagens ERC, van der Peet DL, et al. Differences in Esophageal Cancer Surgery in Terms of Surgical Approach and Extent of Lymphadenectomy: Findings of an International Survey. Ann Surg Oncol 2019;26:2063-72. [Crossref] [PubMed]
- Logan A. The surgical treatment of carcinoma of the esophagus and cardia. 1963(0022-5223 (Print)).
- Altorki N. En-bloc esophagectomy--the three-field dissection. Surg Clin North Am 2005;85:611-9. xi. [Crossref] [PubMed]
- Cuesta MA, Weijs TJ, Bleys RL, et al. A new concept of the anatomy of the thoracic oesophagus: the meso-oesophagus. Observational study during thoracoscopic esophagectomy. Surg Endosc 2015;29:2576-82. [Crossref] [PubMed]
- Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1986;1:1479-82. [Crossref] [PubMed]
- Collard JM, Otte JB, Fiasse R, et al. Skeletonizing en bloc esophagectomy for cancer. Ann Surg 2001;234:25-32. [Crossref] [PubMed]
- Wang H, Tang H, Fang Y, et al. Morbidity and Mortality of Patients Who Underwent Minimally Invasive Esophagectomy After Neoadjuvant Chemoradiotherapy vs Neoadjuvant Chemotherapy for Locally Advanced Esophageal Squamous Cell Carcinoma: A Randomized Clinical Trial. JAMA Surg 2021;156:444-51. [Crossref] [PubMed]
- Cumpston M, Li T, Page MJ, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev 2019;10:ED000142. [Crossref] [PubMed]
- Tierney JF, Stewart LA, Ghersi D, et al. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 2007;8:16. [Crossref] [PubMed]
- Grotenhuis BA, van Heijl M, Zehetner J, et al. Surgical management of submucosal esophageal cancer: extended or regional lymphadenectomy? Ann Surg 2010;252:823-30. [Crossref] [PubMed]
- Donohoe CL, O'Farrell NJ, Ravi N, et al. Evidence-based selective application of transhiatal esophagectomy in a high-volume esophageal center. World J Surg 2012;36:98-103. [Crossref] [PubMed]
- Yekebas EF, Schurr PG, Kaifi JT, et al. Effectiveness of radical en-bloc-esophagectomy compared to transhiatal esophagectomy in squamous cell cancer of the esophagus is influenced by nodal micrometastases. J Surg Oncol 2006;93:541-9. [Crossref] [PubMed]
- Hulscher JB, van Sandick JW, de Boer AG, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the esophagus. N Engl J Med 2002;347:1662-9. [Crossref] [PubMed]
- Johansson J, DeMeester TR, Hagen JA, et al. En bloc vs transhiatal esophagectomy for stage T3 N1 adenocarcinoma of the distal esophagus. Arch Surg 2004;139:627-31; discussion 631-3. [Crossref] [PubMed]
- Rizzetto C, DeMeester SR, Hagen JA, et al. En bloc esophagectomy reduces local recurrence and improves survival compared with transhiatal resection after neoadjuvant therapy for esophageal adenocarcinoma. J Thorac Cardiovasc Surg 2008;135:1228-36. [Crossref] [PubMed]
- Corsini EM, Mitchell KG, Zhou N, et al. Modified En Bloc Esophagectomy Compared With Standard Resection After Neoadjuvant Chemoradiation. Ann Thorac Surg 2021;111:1133-40. [Crossref] [PubMed]
- Hagen JA, Peters JH, DeMeester TR. Superiority of extended en bloc esophagogastrectomy for carcinoma of the lower esophagus and cardia. J Thorac Cardiovasc Surg 1993;106:850-8; discussion 858-9. [Crossref] [PubMed]
- Altorki NK, Girardi L, Skinner DB. En bloc esophagectomy improves survival for stage III esophageal cancer. J Thorac Cardiovasc Surg 1997;114:948-55; discussion 955-6. [Crossref] [PubMed]
- Fu J, Du Z, Zheng C. Total Mesoesophageal Dissection by Thoracoscopy Combined with Laparoscopy in Radical Resection of Esophageal Carcinoma. Chinese Journal of Minimally Invasive Surgery 2012;12:884-7.
- Junhui F, Haosheng Z, Zesen D, et al. Application of Esophageal Cancer Mesoesphagel Excision in the Quality Control of Thoracoscopic Combined Laparoscopic Esophagectomy. Chinese Journal of Minimally Invasive Surgery 2017;17:868-72.
- Fujiwara H, Shiozaki A, Konishi H, et al. Hand-assisted laparoscopic transhiatal esophagectomy with a systematic procedure for en bloc infracarinal lymph node dissection. Dis Esophagus 2016;29:131-8. [Crossref] [PubMed]
- Lin J, He J, Chen S, et al. Outcomes of minimally invasive total mesoesophageal excision: a propensity score-matched analysis. Surg Endosc 2022;36:3234-45. [Crossref] [PubMed]
- Akiyama Y, Iwaya T, Endo F, et al. Thoracoscopic esophagectomy with total meso-esophageal excision reduces regional lymph node recurrence. Langenbecks Arch Surg 2018;403:967-75. [Crossref] [PubMed]
- Biere SS, van Berge Henegouwen MI, Maas KW, et al. Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: a multicentre, open-label, randomised controlled trial. Lancet 2012;379:1887-92. [Crossref] [PubMed]
- Luketich JD, Pennathur A, Awais O, et al. Outcomes after minimally invasive esophagectomy: review of over 1000 patients. Ann Surg 2012;256:95-103. [Crossref] [PubMed]
- Straatman J, van der Wielen N, Cuesta MA, et al. Minimally Invasive Versus Open Esophageal Resection: Three-year Follow-up of the Previously Reported Randomized Controlled Trial: the TIME Trial. Ann Surg 2017;266:232-6. [Crossref] [PubMed]
- Lubbers M, van Det MJ, Kreuger MJ, et al. Totally minimally invasive esophagectomy after neoadjuvant chemoradiotherapy: Long-term oncologic outcomes. J Surg Oncol 2018;117:651-8. [Crossref] [PubMed]
- Warner S, Chang YH, Paripati H, et al. Outcomes of minimally invasive esophagectomy in esophageal cancer after neoadjuvant chemoradiotherapy. Ann Thorac Surg 2014;97:439-45. [Crossref] [PubMed]
- Tsunoda S, Shinohara H, Kanaya S, et al. Mesenteric excision of upper esophagus: a concept for rational anatomical lymphadenectomy of the recurrent laryngeal nodes in thoracoscopic esophagectomy. Surg Endosc 2020;34:133-41. [Crossref] [PubMed]
Cite this article as: Su F, Jiao H, Yin J, Fang Y, Tan L, Shen Y. Effect of en bloc esophagectomy and total meso-esophagectomy on esophageal cancer patients: a systematic review and meta-analysis. AME Med J 2022;7:23.

